Mechanistic Studies of a Skatole-Forming Glycyl Radical Enzyme Suggest Reaction Initiation via Hydrogen Atom Transfer
Abstract
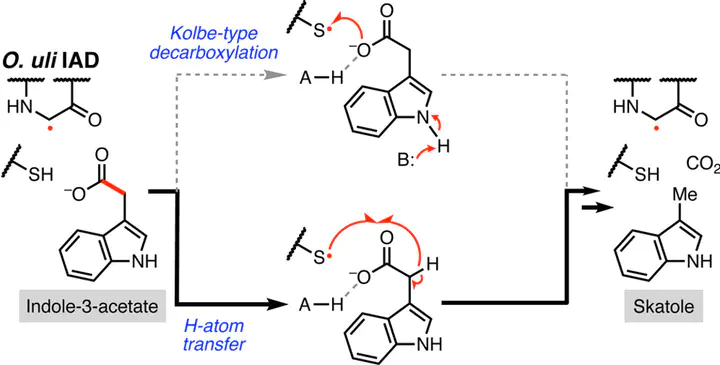
Gut microbial decarboxylation of amino acid-derived arylacetates is a chemically challenging enzymatic transformation which generates small molecules that impact host physiology. The glycyl radical enzyme (GRE) indoleacetate decarboxylase from Olsenella uli (Ou IAD) performs the non-oxidative radical decarboxylation of indole-3-acetate (I3A) to yield skatole, a disease-associated metabolite produced in the guts of swine and ruminants. Despite the importance of IAD, our understanding of its mechanism is limited. Here, we characterize the mechanism of Ou IAD, evaluating previously proposed hypotheses of: (1) a Kolbe-type decarboxylation reaction involving an initial 1-e– oxidation of the carboxylate of I3A or (2) a hydrogen atom abstraction from the α carbon of I3A to generate an initial carbon-centered radical. Site-directed mutagenesis, kinetic isotope effect experiments, analysis of reactions performed in D2O, and computational modeling are consistent with a mechanism involving initial hydrogen atom transfer. This finding expands the types of radical mechanisms employed by GRE decarboxylases and non-oxidative decarboxylases, more broadly. Elucidating the mechanism of IAD decarboxylation enhances our understanding of radical enzymes and may inform downstream efforts to modulate this disease-associated metabolism.