Reversible O–O Bond Scission and O₂ Evolution at MOF-supported Tetramanganese Clusters
Abstract
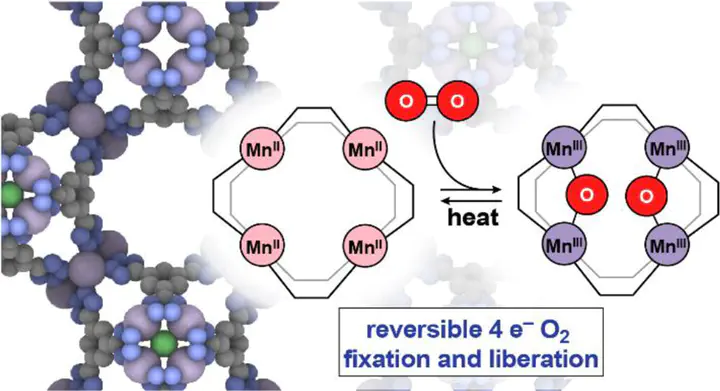
The scission of the O–O bond in O2 and the formation of O–O bonds during the evolution of O2 in photosynthesis are the engines of aerobic life as we know it. Likewise, the reduction of O2 and its reverse, the oxidation of reduced oxygen species to form O2, are indispensable components of emerging renewable technologies for energy storage and conversion. Storing and unleashing the energy contained within the O2 molecule requires control over the formation or scission of the four-electron double bond between two oxygen atoms. Nature performs these demanding multi-electron transformations by distributing the redox burden among multiple metal ions: evolution created metalloenzymes with polynuclear clusters wherein metal ions act in concert to deliver or accept multiple electrons. Most relevant in this sense are the tetramanganese-calcium (Mn4CaO5) cluster in the oxygen-evolving complex of photosystem II, responsible for O2 formation, and the tricopper clusters in multi-copper oxidases, which mediate O2 reduction. The critical step in both O2 reduction and O2 evolution is the interconversion between multinuclear metal-oxo species, where the O atoms carry formal –2 charges, and O–O bonded species where the oxygen atoms are more oxidized. Attempts to mimic the natural systems with synthetic analogs have led to a number of elegant molecular clusters that shed light on the electron transfer events and stepwise mechanism of O–O bond cleavage and formation. To our knowledge, however, the interconversion between molecular O2 and metal-oxo species in either synthetic or enzymatic discrete systems has not been documented. Here, we report that a tetramangenese cluster formed by self-assembly within a metal–organic framework (MOF) spontaneously cleaves the oxygen-oxygen double bond and reduces O2 by four electrons at room temperature. The ensuing tetranuclear manganese-oxo cluster engages in weak C–H bond activation and, more importantly, is competent for O–O bond formation and O2 evolution at elevated temperature, enabled by the head-to-head orientation of two fully reduced oxo atoms that bridge neighboring Mn pairs. This study demonstrates the viability of four-electron interconversion between molecular O2 and metal-oxo species and highlights the importance of site-isolation for achieving multi-electron chemistry at polynuclear metal clusters.