Abstract
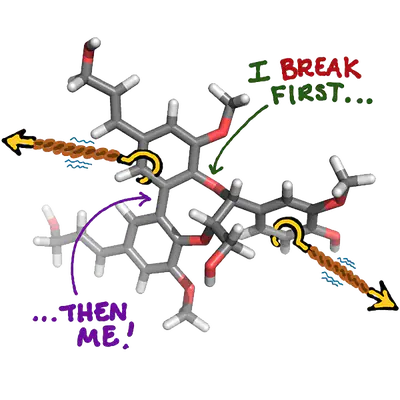
The directed depolymerization of lignin biopolymers is of utmost relevance for the valorization or commercialization of biomass fuels. We present a computational and theoretical screening approach to identify potential cleavage pathways and resulting fragments that are formed during depolymerization of lignin oligomers containing two to six monomers. We have developed a chemical discovery technique to identify the chemically relevant putative fragments in eight known polymeric linkage types of lignin. Obtaining these structures is a crucial precursor to the development of any further kinetic modeling. We have developed this approach by adapting steered molecular dynamics calculations under constant force and varying the points of applied force in the molecule to diversify the screening approach. Key observations include relationships between abundance and breaking frequency, the relative diversity of potential pathways for a given linkage, and the observation that readily cleaved bonds can destabilize adjacent bonds, causing subsequent automatic cleavage.